 To avoid these misconceptions, it is important to introduce, and emphasise, the correct use of the terms ion and molecule from early on in a student’s chemical studies. Common misconceptionsĪs the students develop their understanding of chemical bonding further, it is common for students to refer to ionic compounds as molecules or to refer to intermolecular forces when explaining properties of ionic compounds. Gold atoms cannot be broken down into anything smaller while still retaining the properties. For example, a gold coin is simply a very large number of gold atoms molded into the shape of a coin, with small amounts of other, contaminating elements. You can download instructions, an example grid and game cards below. An atom is the smallest unit of matter that retains all of the chemical properties of an element. The terms molecule, compound, and atom can be confusing Heres an explanation of what a molecule is (and is not) with examples of common molecules. One such game is based on the classic Connect 4 game. Using games and an element of competition can be helpful to bring some variety to the necessary student practice. Together, the number of protons and the number of neutrons determine an element’s mass number: mass number protons + neutrons. 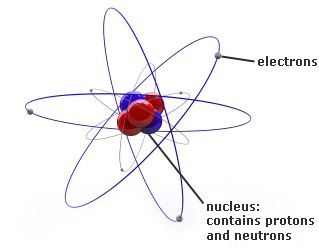
Owing to the interweaving of the terms atom, ion and molecule when describing the different particles, it is unsurprising that students get confused. Introducing the electron now, before students meet the other sub-atomic particles, can help to embed the idea that the loss of electrons results in a positively charged ion, and may help reduce confusion later on. 
This latter point is something students often struggle with later on in their studies. When an atom/molecule loses negatively charged electron(s), a positive ion is formed. When an atom/molecule gains negatively charged electron(s), a negative ion is formed. However it may be worth introducing students to the electron at this point. At this level students only need to know that an ion is a positively or negatively charged particle. Venn diagrams help students organise their understanding of the different particle types, as described in Atoms, elements, molecules, compounds and mixtures (rsc.li/2wzLsxS).Īn atom or a molecule can lose or gain electron(s) to form an ion. Use of colour helps to distinguish between the atom types further. For example, all hydrogen atoms, and only hydrogen atoms, contain one proton and have an atomic number of 1. In fact even Dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (Figure 1). 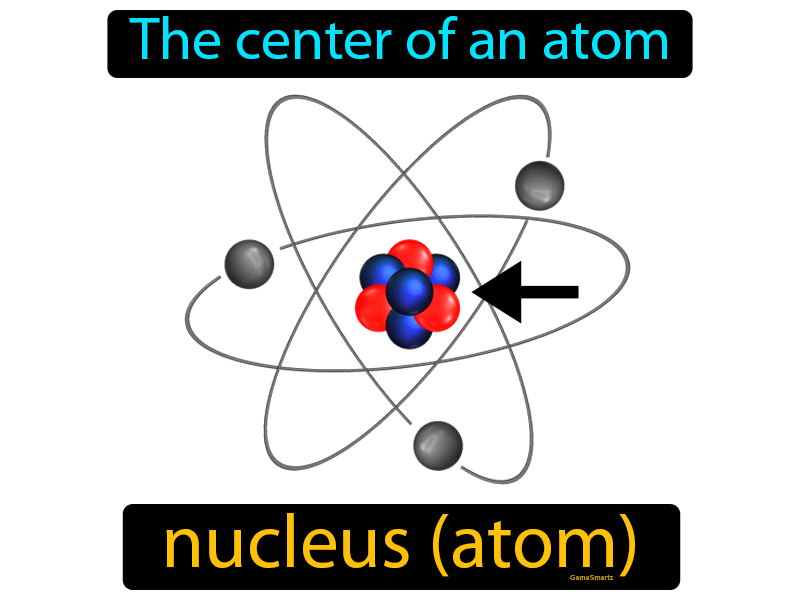
Particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
